On Tuesday, the U.S. Food and Drug Administration (FDA) authorized the first at-home diagnostic test for COVID-19 under an Emergency Use Authorization (EUA).
The testing kit, named Pixel, is developed by LabCorp. LabCorp, a leading global life sciences company focused on improving health by diagnoses, has been around for over 40 years.
“LabCorp continues to develop new ways to help patients and healthcare providers fight the COVID-19 crisis through our leading testing capabilities and deep scientific and research expertise,” said Adam Schechter, the president and CEO of LabCorp.
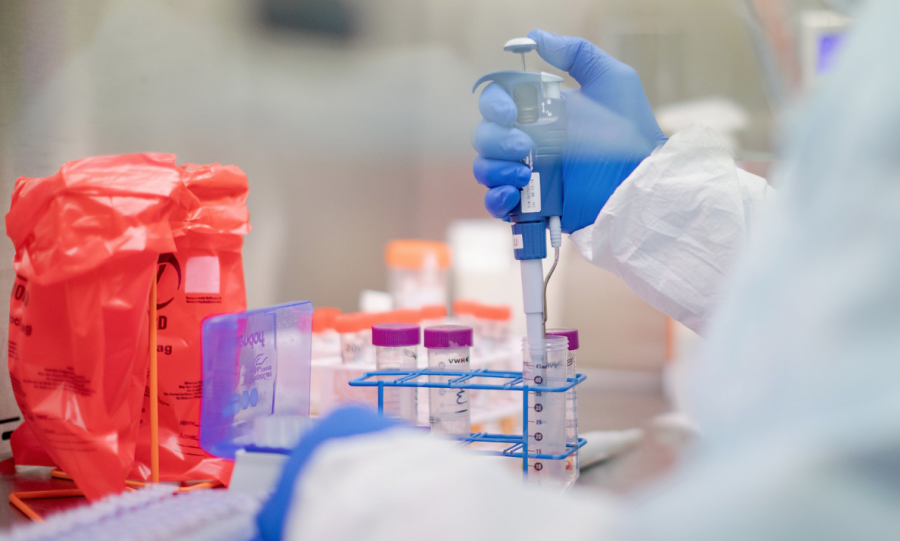
 To buy a testing kit, consumers must complete an eligibility survey first. If it is approved, a testing kit will be shipped to their preferred location. Then, consumers will collect a nasal sample and ship it back, where the sample will be tested in a world-class lab. Once the results have been processed, consumers will receive an email about their results.
To buy a testing kit, consumers must complete an eligibility survey first. If it is approved, a testing kit will be shipped to their preferred location. Then, consumers will collect a nasal sample and ship it back, where the sample will be tested in a world-class lab. Once the results have been processed, consumers will receive an email about their results.
Due to the limited supply, the Pixel is currently only available to frontline healthcare workers and first responders who may have COVID-19 symptoms or may have been exposed to it, as suggested by the guidelines set by the Centers for Disease Control and Prevention (CDC). However, LabCorp plans to make the Pixel available to consumers in the coming weeks.
With the Pixel, LabCorp hopes to minimize the risk of infection to frontline workers and reduce the demand for personal protective equipment (PPE).
“Our at-home collection kits are designed to make it easier and safer to test healthcare workers and first responders during this important time,” Schechter said.